Metal
In chemistry, a metal is an element that readily forms positive ions (cations) and has metallic bonds. Metals are sometimes described as a lattice of positive ions surrounded by a cloud of delocalized electrons. The metals are one of the three groups of elements as distinguished by their ionization and bonding properties, along with the metalloids and nonmetals. On the periodic table, a diagonal line drawn from boron (B) to polonium (Po) separates the metals from the nonmetals. Most elements on this line are metalloids, sometimes called semi-metals; elements to the lower left are metals; elements to the upper right are nonmetals. A modern definition of metals is that they have overlapping conduction bands and valence bands in their electronic structure. This definition opens up the category for metallic polymers and other organic metals, which have been made by researchers and employed in high-tech devices. These synthetic materials often have the characteristic silvery-grey reflectiveness of elemental metals. The traditional definition focuses on the bulk properties of metals. They tend to be lustrous, ductile, malleable, and good conductors of electricity, while nonmetals are generally brittle (for solid nonmetals), lack lustre, and are insulators.
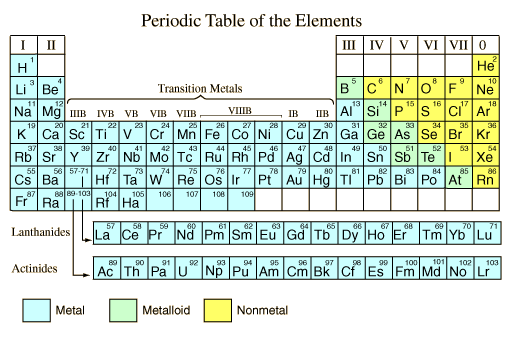
In the periodic table, you can see a stair-stepped line starting at Boron (B), atomic number 5, and going all the way down to Polonium (Po), atomic number 84. Except for Germanium (Ge) and Antimony (Sb), all the elements to the left of that line can be classified as metals. These metals have properties that you normally associate with the metals you encounter in everyday life:
- They are solid (with the exception of mercury, Hg, a liquid).
- They are shiny, good conductors of electricity and heat.
- They are ductile(they can be drawn into thin wires).
- They are malleable(they can be easily hammered into very thin sheets).
All these metals tend to lose electrons easily. The following figure shows the metals.
Non Metals
As far as elements are concerned, a nonmetal is simply an element that does not display the properties of a metal. It is not defined by what it is, but by what it is not. It doesn’t look metallic, can’t be drawn into a wire or pounded into shape or bent, doesn’t conduct heat or electricity well, and doesn’t have a high melting or boiling point.
The nonmetals are in the minority on the periodic table, mostly pushed to the right hand side of the periodic table.
The exception is hydrogen, which behaves as a nonmetal at room temperature and pressure and is found on the upper left corner of the periodic table. Under conditions of high pressure, hydrogen is predicted to behave as an alkali metal.
Here’s a look at which elements are nonmetals, how to locate the nonmetals on the table, and their common properties.
LOCATION ON THE NONMETALS ON THE PERIODIC TABLE
The nonmetals are located on the upper right side of the periodic table. Nonmetals are separated from metals by a line that cuts diagonally through the region of the periodic table containing elements with partially filled p orbitals. The halogens and noble gasesare nonmetals, but the nonmetal element group usually is considered to consist of the following elements:
- hydrogen
- carbon
- nitrogen
- oxygen
- phosphorus
- sulfur
- selenium
The halogen elements are:
- fluorine
- chlorine
- bromine
- iodine
- astatine
- Possibly element 117 (tennessine), although most scientists think this element will behave as a metalloid.
The noble gas elements are:
- helium
- neon
- argon
- krypton
- xenon
- radon
- element 118 – oganesson (predicted to be a liquid, but still a nonmetal)
PROPERTIES OF NONMETALS
Nonmetals have high ionization energies and electronegativities. They are generally poor conductors of heat and electricity. Solid nonmetals are generally brittle, with little or no metallic luster.
Most nonmetals have the ability to gain electrons easily. Nonmetals display a wide range of chemical properties and reactivities.
SUMMARY OF COMMON PROPERTIES
- High ionization energies
- High electronegativities
- Poor thermal conductors
- Poor electrical conductors
- Brittle solids – not malleable or ductile
- Little or no metallic luster
- Gain electrons easily
- Dull, not metallic-shiny, although they may be colorful
- Lower melting points and boiling point than the metals
COMPARING THE METALS AND NONMETALS
Here’s a comparison of the physical and chemical properties of the metals and nonmetals. These properties apply to the metals in general (alkali metals, alkaline earth, transition metals, basic metals, lanthanides, actinides) and nonmetals in general (nonmetals, halogens, noble gases).
| Metals | Nonmetals | |
| chemical properties | easily lose valence electrons | easily share or gain valence electrons |
| 1-3 electrons (usually) in the outer shell | 4-8 electrons in the outer shell (7 for halogens and 8 for noble gases) | |
| form basic oxides | form acidic oxides | |
| good reducing agents | good oxidizing agents | |
| have low electronegativity | have higher electronegativity | |
| physical properties | solid at room temperature (except mercury) | may be liquid, solid, or gas (noble gases are gases) |
| have metallic luster | do not have metallic luster | |
| good conductor of heat and electricity | poor conductor of heat and electricity | |
| typically malleable and ductile | usually brittle | |
| opaque in a thin sheet | transparent in a thin sheet |